Project: C08
Dysregulation of Iron homeostasis-induced cellular inflammatory and neuro-degenerative processes in glaucoma
Glaucoma is the second most common cause of irreversible blindness worldwide and is characterized by progressive loss of retinal ganglion cells (RGC). While elevated intraocular pressure (IOP) is a well-known risk factor and IOP lowering a well-acknowledged treatment, RGC loss can proceed despite sufficient IOP reduction. This suggests that once a vicious circle has been initiated, it is maintained independently of IOP control.
We and others have shown that glaucoma goes along with significant neuroinflammation and microglial activation. The major hypothesis of this grant application is that there is an interplay of glaucoma and neuroinflammation being triggered amongst others by a disbalance of iron homeostasis. The iron imbalance is the most probable cause leading to activation of the originally resting microglia. Therefore, the disease of glaucoma continues due to the activated microglia, albeit intraocular pressure is being treated.
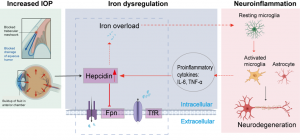
The first objective will be to further investigate the sequence of IOP elevation and neuroinflammation through IOP triggered hepcidin-iron dysregulation. Special focus will be put on microglial activation, pro-inflammatory cytokine secretion and ferroptosis due to iron overload and increased IOP in vivo, retinal explants and cell culture experiments.
The second objective will translate those findings into the human context through analysis of aqueous humor, serum, retina, and optic nerve tissue from glaucoma patients.
The third objective will define the pathophysiological signaling mechanisms of glaucoma by in vivo and in vitro models with a genetic deficiency of the BMP type I receptors Alk2 and Alk3 that are characterized by protection to induce inflammatory parameters or hepcidin and expose these models to intraocular pressure.
Key Methods:
• In vivo glaucoma models with IOP elevation and genetically modified mice
• Retinal explant cultures
• Microglia-focused cell culture models
• Iron overload and ferroptosis assays
• ELISA-based cytokine and hepcidin analyses
• qPCR-based gene expression analyses
• Immunohistochemical / immunofluorescence-based protein analyses
• Oxidative stress and ROS measurements
• Analysis of the hepcidin–ferroportin and BMP signaling pathways
• Translational analyses of human samples (aqueous humor, serum, retina, optic nerve)


